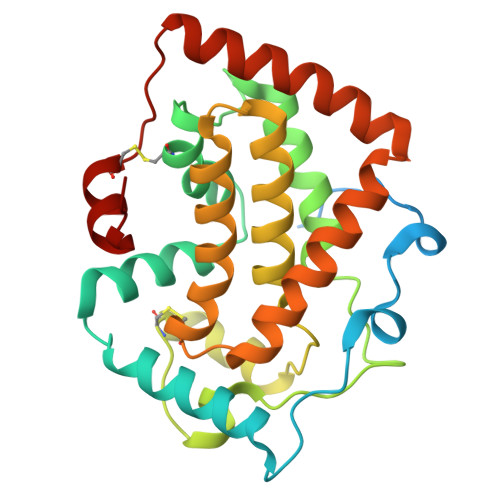
Thermotolerant class A acid phosphatase active across broad pH range and diverse substrates.
Recio, M.I., Gavira, J.A., de La Torre, J., Cano-Munoz, M., Martinez-Rodriguez, S., Daddaoua, A., Duque, E., Ramos, J.L.(2025) Protein Sci 34: e70244-e70244
- PubMed: 40815342
- DOI: https://doi.org/10.1002/pro.70244
- Primary Citation of Related Structures:
9HTZ - PubMed Abstract:
M2-32 is a non-specific acid phosphatase with a rare ability to function across a broad pH range (3.5-8.5). Analysis using SWISS-PROT Prf Profiles classifies it as a class A acid phosphatase (Z-score: 78.97), sharing 50%-60% sequence similarity with enzymes such as PhoC and PhoN. For detailed characterization, the gene encoding M2-32 was cloned into the pET28(b) vector, overexpressed in Escherichia coli BL21 (DE3), and subsequently purified. Although the monomeric form of M2-32 has a molecular weight of ~28 kDa, size exclusion chromatography, dynamic light scattering, and sedimentation studies revealed a dimeric form in solution. Enzymatic assays using p-nitrophenyl phosphate, 4-methylumbelliferyl phosphate, 3'-and 5'-adenosine monophosphate demonstrated robust activity over a pH range of 4.0-8.0 at both 30 and 50°C. Differential scanning fluorimetry indicated an unfolding temperature close to 47°C; however, the enzyme refolded after heat denaturation at 80°C. We have determined the x-ray crystal structure of M2-32 by molecular replacement using an AlphaFold2-guided truncated model, achieving a resolution of 2.2 Å. The protein crystallized as a dimer-of-dimers. Each monomer (residues 38-274) adopts an all-alpha-helical fold composed of 14 helices and two disulfide bonds. Docking studies with adenosine monophosphates, combined with site-directed mutagenesis, identified His174, Arg207, His213, Asp217 as critical catalytic residues, and Tyr136 and Ser172 probably involved in substrate recognition. Mutations at these positions resulted in over 90% loss of enzymatic activity, highlighting their functional significance.
- Estación Experimental del Zaidín, Consejo Superior de Investigaciones Científicas, Granada, Spain.
Organizational Affiliation: