Positioning of Nuclear Localization Signals as an Evolutionary Marker in Polyomavirus Large Tumor Antigens
Hoad, M., Pavan, S., Nematollahzadeh, S., Tietz, O., Reeman, J., Forwood, J.K., Alvisi, G.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Importin subunit alpha-1 | 510 | Mus musculus | Mutation(s): 0 Gene Names: Kpna2, Rch1 | 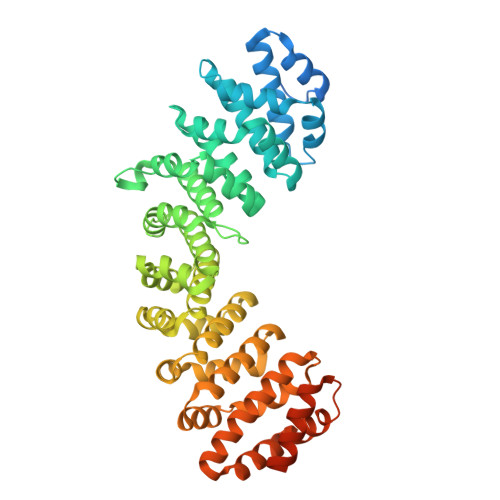 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P52293 (Mus musculus) Explore P52293 Go to UniProtKB: P52293 | |||||
IMPC: MGI:103561 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P52293 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by: Sequence | 3D Structure
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Truncated large T antigen | 24 | Black sea bass polyomavirus 1 | Mutation(s): 0 EC: 5.6.2.4 |  | |
UniProt | |||||
Find proteins for A0A0A1C723 (black sea bass-associated polyomavirus 1) Explore A0A0A1C723 Go to UniProtKB: A0A0A1C723 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0A1C723 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 78.802 | α = 90 |
| b = 89.188 | β = 90 |
| c = 99.659 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Coot | model building |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Not funded | -- |