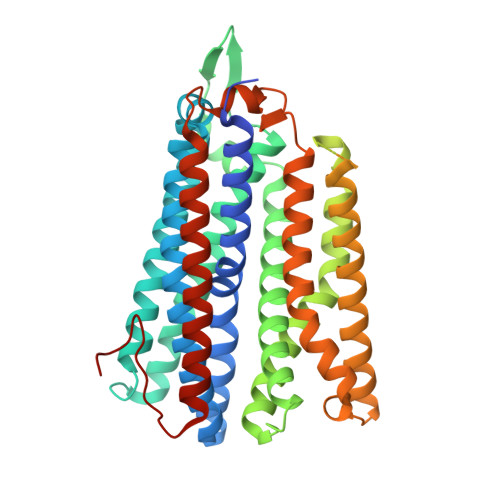
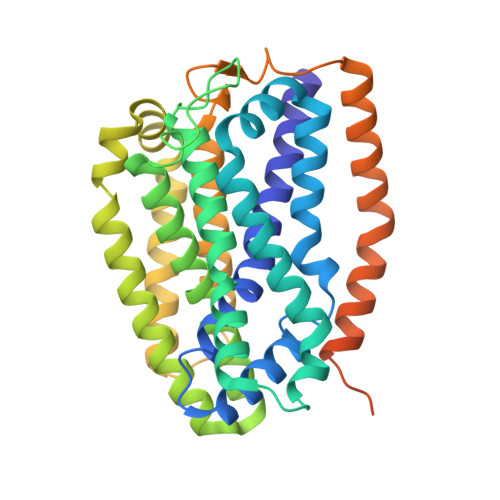
The Mycobacterium smegmatis bd -II terminal oxidase employs a carboxylate shift mechanism.
Kovalova, T., Janczak, M., Gamiz-Hernandez, A.P., Lundin, D., Sharma, S., Vilhjalmsdottir, J., Sjostrand, D., Kaila, V.R.I., Hogbom, M., Adelroth, P.(2026) Proc Natl Acad Sci U S A 123: e2515348123-e2515348123
- PubMed: 41805574
- DOI: https://doi.org/10.1073/pnas.2515348123
- Primary Citation Related Structures:
9R2G - PubMed Abstract:
Cytochrome bd is a terminal oxidase expressed under low oxygen conditions and central for the survival of many pathogens. Here, we characterize the cyt bd -II from Mycobacterium smegmatis, a member of a hitherto uncharacterized evolutionary group (qOR-2) of bd oxidases, by combining biochemical studies with cryo-electron microscopy (cryo-EM), and multiscale simulations. Overexpressing the appCB operon in its native host led to production of a highly active bd -II ( k obs = 30 e - s -1 ) that together with a high-resolution (2.8 Å) cryo-EM structure and multiscale simulations reveal unique proton pathways and oxygen channels responsible for its function. We propose that a pH-dependent molecular switch, involving coordination changes of heme d and surrounding bulky residues regulate substrate access into the active site. Taken together, our findings provide detailed mechanistic insight of qOR-2 type bd oxidases, and a basis for understanding the evolution of the superfamily.
- Department of Biochemistry and Biophysics, Stockholm University, Stockholm 116 91, Sweden.
Organizational Affiliation: