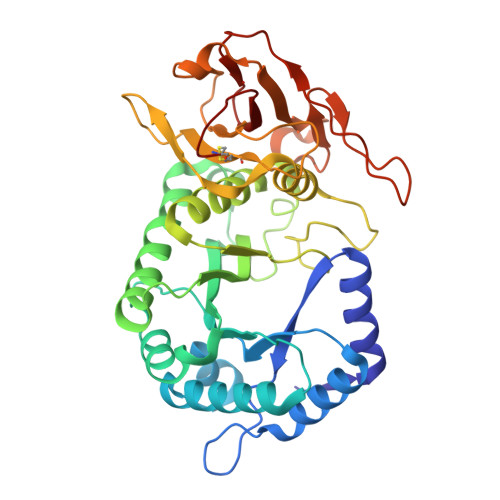
Fragment Screening and Structure-Guided Development of Heparanase Inhibitors Reveal Orthosteric and Allosteric Inhibition.
Davies, L.J., Whitefield, C., Kim, H., Nitsche, C., Jackson, C.J., Frkic, R.L.(2026) ACS Med Chem Lett 17: 383-390
- PubMed: 41704381
- DOI: https://doi.org/10.1021/acsmedchemlett.5c00587
- Primary Citation Related Structures:
9O1R, 9O1S, 9O1T, 9O1U, 9O1V, 9O1W, 9O1X, 9O1Y, 9O1Z, 9O20, 9O21, 9O22, 9O23, 9O24, 9O25, 9O26, 9O27, 9O28, 9O29, 9O2A, 9O2B, 9O2C, 9O2D, 9O2E, 9O2F, 9O2G, 9O2H, 9O2I, 9O2J, 9O2K, 9O2L, 9O2M - PubMed Abstract:
Heparanase is the sole enzyme responsible for breaking down heparan sulfate within the extracellular matrix, and its overexpression is linked to human diseases. Despite heparanase being a promising drug target, most efforts have focused on substrate mimetics, which have failed clinical trials, highlighting the need for new inhibitor scaffolds. Here, we employed fragment-based drug design to explore a novel chemical space to develop small molecule inhibitors of heparanase. We used a crystallographic and computational approach to identify 31 fragments that bind heparanase; five of these inhibited heparanase in the micromolar range. One of these fragments underwent two cycles of fragment growing, which resulted in a compound with a 7-fold increased potency compared to the initial hit. The results from our fragment screen unveil untapped chemical space for heparanase inhibition, paving the way for the development of potent drug leads with the potential to transform the treatment of heparanase-related diseases.
- Research School of Chemistry, Australian National University, Canberra, ACT 2601, Australia.
Organizational Affiliation: